- India
- International
Corbevax gets approval as first mix-and-match booster vaccine
Biological E: Trial showed significant enhancement in immune response
 So far, mixing and matching Covid-19 vaccines was not allowed in India and the third dose had to be of the same vaccine used for the first and second doses. (Express Photo/File)
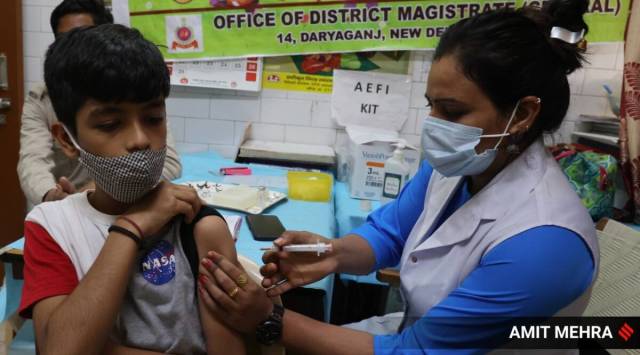
So far, mixing and matching Covid-19 vaccines was not allowed in India and the third dose had to be of the same vaccine used for the first and second doses. (Express Photo/File)Vaccine manufacturer Biological E’s protein sub-unit Covid-19 vaccine Corbevax has become the first to be approved by the country’s drug regulator as a heterologous booster in adults, meaning those who have received Covishield or Covaxin as their first or second dose can take it as a third booster shot.
So far, mixing-and-matching of Covid-19 vaccines was not allowed in India and the third dose had to be the same vaccine used for the first and second dose.
With the Drug Controller General of India (DCGI) now approving the heterologous booster, the government will have to take a call on whether it should be included in the country’s free vaccination drive.
India has so far administered 193.9 crore doses of vaccine in total, of which 101 crore are first doses, 89 crore second doses, and 3.5 crore precaution doses, according to data from the government’s CoWIN portal.
In a statement Saturday, the Hyderabad-based company said, “BE’s clinical trial data showed that CORBEVAX® booster dose provided significant enhancement in immune response and excellent safety profile required for an effective booster.”

🚨 Limited Time Offer | Express Premium with ad-lite for just Rs 2/ day 👉🏽 Click here to subscribe 🚨
The approval came after a trial involving 416 people who were administered Corbevax or placebo (an agent with no therapeutic value) six months after having received two doses of either Covaxin or Covishield.
The company, in its statement, said the levels of neutralising antibodies – antibodies that specifically attack or block the Sars-CoV-2 virus – increased significantly as compared to the placebo.
After the booster dose of Corbevax, neutralising antibodies against Omicron were found in 91% of those who had received Covishield earlier and 75% of those who had received Covaxin.
Not just antibody levels, the company also found cell-mediated immunity to be higher as compared to the placebo. “The Corbevax heterologous booster vaccine was well tolerated and safe. There were no severe or adverse events of interest for 3 months of follow-up after the booster dose was administered,” the company said.
Currently, the vaccine is in use for immunisation of children between the ages of 12 and 14 years under the government programme. It is also available on payment for children between 12 and 17 years at private vaccination centres.
The company said that so far 51.7 million doses of the vaccine had been administered to children across the country, with 100 million doses having been supplied to the government.
Apr 26: Latest News
- 01
- 02
- 03
- 04
- 05